Understanding the Spectrum of Skin Failure
Stevens-Johnson Syndrome (SJS) and Toxic Epidermal Necrolysis (TEN) are rare, life-threatening emergencies where your immune system overreacts so violently that your skin begins to shed. Think of it as an extreme version of a burn injury, except the damage comes from within. First described in 1922 by Albert Mason Stevens and Frank Chambliss Johnson, these conditions were once thought to be separate diseases. Today, doctors understand they exist on a single continuum. The difference simply depends on how much skin is involved.
If you have ever heard of someone surviving near-total skin peeling due to medication, you have likely encountered stories about Toxic Epidermal Necrolysis. It is critical to recognize early warning signs because the margin for error is incredibly small. Mortality rates can range from 5% for milder cases to nearly 25% or higher in severe instances. Understanding this distinction isn't just academic; for a patient or caregiver, spotting the flu-like symptoms before the rash appears could save a life.
The Difference Between SJS and TEN
The medical community classifies these conditions based on the percentage of body surface area (BSA) where the skin detaches. A helpful way to visualize BSA is using the "rule of nines," commonly used in burns, where different body parts represent specific percentages of total skin. In the case of SJS and TEN, the cutoff points define the diagnosis:
- Stevens-Johnson Syndrome: Less than 10% of skin surface detached.
- SJS-TEN Overlap: Between 10% and 30% detachment.
- Toxic Epidermal Necrolysis: More than 30% of skin surface detached.
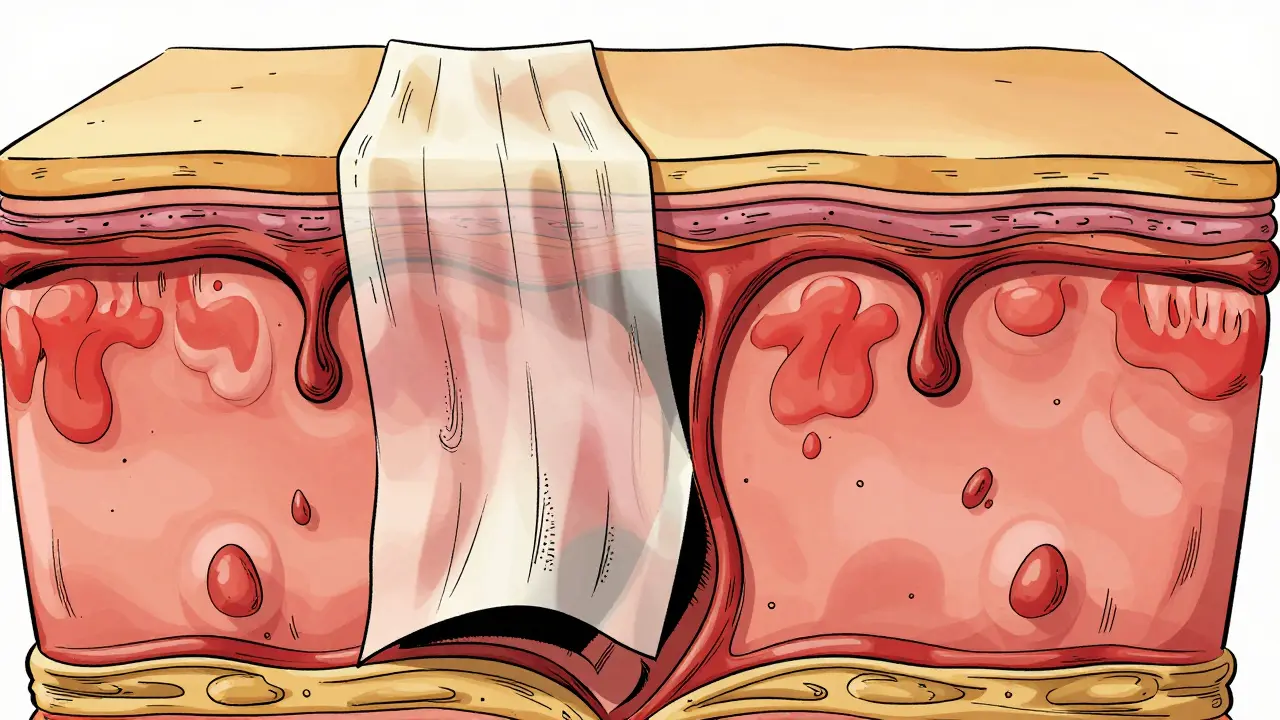
This classification matters significantly for prognosis. As the amount of detached skin increases, so does the risk of infection, fluid loss, and death. The skin acts as our primary barrier against bacteria. When that barrier fails across a large portion of the body, the body loses fluids and electrolytes rapidly, similar to a severe burn victim. This process creates raw, weeping surfaces that invite dangerous infections leading to sepsis and multi-organ failure.
Clinical Signs and Symptom Timeline
The progression of SJS/TEN follows a distinct pattern that rarely happens overnight. It typically starts with a prodromal phase lasting a few days. During this stage, patients often feel they have the flu. You might experience high fevers exceeding 38.9°C (102°F), headaches, a sore throat, coughing, and general malaise. Eye irritation is common here too, mimicking conjunctivitis.
Within one to three days, the cutaneous phase begins. The skin changes start as flat, red, or purple spots (macules) that often begin on the trunk and spread outward. These lesions quickly evolve into blisters and large sheets of skin separation. A key clinical sign doctors look for is a positive Nikolsky sign. If gentle pressure on the skin causes the top layer to slide off, it indicates significant adhesion breakdown between the epidermis and dermis.
Mucosal involvement is universal in these cases. Nearly every patient develops severe issues inside the mouth, eyes, and genitalia. About 90% of patients suffer oral ulcers making swallowing painful. The eyes are particularly vulnerable; inflammation here can lead to permanent vision problems if not managed carefully by an ophthalmologist immediately.

What Triggers the Reaction?
While infections like Mycoplasma pneumoniae can trigger this reaction, especially in children, medications are the culprit in over 80% of adult cases. Identifying the specific agent is crucial for preventing recurrence. Certain drug classes carry higher risks than others.
| Drug Class | Specific Agents | Risk Context |
|---|---|---|
| Antiepileptics | Carbamazepine, Phenytoin, Lamotrigine | ~30% of cases; highest risk in first weeks |
| Antibiotics | Sulfonamides (Bactrim), Penicillins | ~20% of cases |
| Gout Medication | Allopurinol | High dose or renal impairment increases risk |
| NSAIDs | Oxicams, Nimesulide | Late-onset presentations possible |
Beyond the specific drugs, genetics play a massive role in who reacts and who doesn't. Some people possess specific genetic markers known as HLA alleles that dramatically increase susceptibility. For instance, carrying the HLA-B*15:02 allele increases the risk of carbamazepine-induced SJS by a staggering 1,000-fold in certain Asian populations. Similarly, the HLA-B*58:01 allele strongly predicts reactions to allopurinol across multiple ethnic groups. Because of this, countries like Taiwan have implemented national screening programs. Testing for these markers before starting medication can prevent the condition entirely.
Diagnosis and Prognostic Scoring
Time is tissue. Diagnosing SJS/TEN requires combining clinical presentation with histology. A skin biopsy remains the gold standard. Pathologists look for full-thickness necrosis of the epidermis with minimal inflammation in the deeper layers of the skin. While waiting for lab results, doctors rely heavily on physical exam findings and the RegiSCAR criteria, which assess acute onset, skin tenderness, and mucosal involvement.
Once admitted, medical teams calculate a mortality risk score called SCORTEN (Score for Toxic Epidermal Necrolysis). This tool evaluates seven factors during the first 24 hours of hospitalization:
- Age over 40 years.
- Presence of malignant cancer.
- Heart rate faster than 120 beats per minute.
- Initial skin detachment greater than 10%.
- Blood urea levels above 10 mmol/L.
- Blood glucose levels above 14 mmol/L.
- Blood bicarbonate levels below 20 mmol/L.
Each factor present adds approximately a 1.5-fold increase in the likelihood of death. Patients with five or more factors face a grim outlook with predicted mortality nearing 90%. Early identification of these markers helps triage care intensity, ensuring resources focus on those most at risk.
Treatment Strategies and Controversies
The cornerstone of treatment is immediate discontinuation of the offending drug. Delaying this step worsens outcomes significantly. Beyond stopping the medication, management focuses on supportive care similar to burn unit protocols. Patients require aggressive fluid resuscitation-often three to four times normal maintenance volumes-to replace losses from the raw skin surface.
Wound care involves using non-adherent dressings to protect the fragile underlying dermis without tearing fresh skin upon removal. Eye care is equally vital, requiring daily specialist review to prevent symblepharon (adhesions) and scarring.
There is ongoing debate regarding immunomodulatory therapies. Historically, Intravenous Immunoglobulin (IVIG) was popular, showing promise in early studies. However, later randomized controlled trials failed to show a clear mortality benefit. Corticosteroids remain controversial because high doses can increase infection risk, yet some centers still use pulse-dose methylprednisolone very early in the course. More recently, cyclosporine has shown potential; a 2016 trial suggested it reduced mortality rates significantly when given early. Additionally, newer agents like etanercept (a TNF-alpha inhibitor) showed zero mortality in small studies when administered within 48 hours of admission. The medical community continues to weigh the balance between suppressing the immune response and keeping the patient safe from secondary infections.
Long-Term Consequences
Surviving the acute phase is only half the battle. Long-term sequelae affect quality of life drastically. About 60% to 80% of survivors report chronic complications. Ocular issues are among the most debilitating. Dry eye syndrome, photophobia, and corneal scarring are common, sometimes leading to legal blindness. Chronic skin issues include pigmentation changes and nail dystrophy, where fingernails fail to regrow correctly.
Genitourinary complications can also arise, such as urethral strictures or vaginal adhesions, requiring surgical correction. The psychological toll should not be overlooked either. Many survivors develop post-traumatic stress disorder (PTSD) stemming from the traumatic ICU experience. Pain management and mental health support become part of the long-term rehabilitation plan alongside wound care.
Prevention and Future Directions
Prevention is far superior to cure in these scenarios. Pharmacogenomic testing is shifting practice standards. We now know that screening for HLA-B*58:01 before prescribing allopurinol reduces incidence significantly. Regulatory bodies like the FDA are pushing for point-of-care tests to speed up this process. Previously, genetic results took weeks; modern tests provide answers in hours, allowing safe prescription decisions immediately.
Patients with a history of SJS or TEN must avoid the culprit drug forever. Even trace amounts can cause re-exposure reactions, which are often more severe and rapid than the initial episode. Medical alerts and tattoos identifying these allergies can be lifesaving measures for emergency personnel unaware of the history.
Can SJS go away on its own?
No, SJS and TEN will not resolve on their own safely. While mild forms of allergic reactions might self-limit, SJS/TEN progresses rapidly to skin detachment. Without intervention, fluid loss and infection lead to high mortality. Immediate hospitalization is required to stop the causative agent and manage wounds.
What are the earliest symptoms to watch for?
The earliest signs mimic the flu. Watch for high fever, sore throat, pain behind the eyes, and malaise occurring shortly after starting a new medication. If these symptoms appear followed by a skin rash, seek emergency care immediately.
Is there a blood test for SJS/TEN?
There is no single blood test for the active disease itself, though labs often show abnormalities in white blood cell counts or chemistry. Diagnosis relies on clinical exam and skin biopsy. However, blood tests are essential for genetic screening (like HLA typing) to assess risk before taking high-risk drugs.
How do doctors determine the severity?
Doctors use the SCORTEN score, assessing age, heart rate, cancer status, fluid chemistry, and extent of skin detachment. This determines the patient's risk of mortality and guides the intensity of ICU care needed.
Are there vaccines for this condition?
No vaccines exist currently. Prevention relies on avoiding specific drugs based on genetic susceptibility and careful monitoring of new prescriptions. Since infections like Mycoplasma can trigger it, general respiratory hygiene helps reduce exposure to infectious triggers.




Goodwin Colangelo
The prodromal phase is really something nobody thinks about until it happens. Most people just assume they have the flu when they feel sick after starting a new drug. This delay in recognizing signs is what makes mortality rates so high in severe cases. If you notice a sore throat followed by rashes you need to act fast. Early removal of the trigger medication is the single biggest factor for survival. We need more public awareness campaigns about these specific warning signs. It saves lives when caregivers spot the difference between normal allergies and this condition. Keeping an eye on mucosal involvement is also critical for early detection. Doctors often miss the Nikolsky sign until it is too late for proper intervention. Prevention through genetic testing is available and should be utilized more widely. Everyone deserves to know their risk profile before taking prescription meds.
Beth LeCours
This looks scary.
Will Baker
Fascinating how we can die just from taking a pill to help us. Thanks for scaring everyone who just got prescribed antibiotics today. It really puts everything in perspective doesn't it. Just another way for modern medicine to ruin your peace of mind.
Dee McDonald
You need to stop making light of life threatening conditions like this. People are dying because of this exact attitude you are showing right now. Your sarcasm does nothing to help anyone who is actually suffering. Read the facts instead of mocking the situation for your own entertainment value. We are trying to save lives here not provide a comedy club for you.
Sam Hayes
I hope you stay safe too and always check your meds carefully glad we are discussing this topic openly
Divine Manna
It is important to understand that the pathology described involves distinct mechanisms. The genetic component specifically regarding HLA alleles is frequently overlooked by laypeople. Medical professionals must prioritize these markers before prescribing high-risk agents. Without such screening patients remain vulnerable to catastrophic epidermal detachment. Furthermore the distinction between drug classes matters significantly for prevention strategies. Many individuals mistakenly assume that older medications carry higher risks than newer alternatives. This assumption fails to account for individual genetic susceptibility patterns found in specific populations. Consequently population-based screening programs have become standard practice in certain nations. The implementation of rapid point-of-care testing drastically reduces the latency period before treatment. Timely intervention remains the single most effective predictor of survival in acute phases. Physicians must also consider the overlap syndrome classification carefully during admission triage. Ignoring the fluid loss dynamics leads to secondary shock complications within twenty-four hours. Wound management protocols differ significantly from standard burn unit approaches regarding adhesion. Ocular involvement represents the most debilitating long-term consequence for survivors. Therefore comprehensive care extends well beyond the initial survival window into rehabilitation.
Joseph Rutakangwa
good summary but misses cost barriers
Joey Petelle
While the science is quaint the real tragedy is our healthcare systems failing the privileged few who need access. One must wonder if the masses even get tested for these markers or just get lucky. The elitism of genetic testing is staggering when poverty prevents basic care. Perhaps we should focus on universal access rather than niche pharmacogenomics. The narrative focuses heavily on technology neglecting social determinants of health outcomes. It is quite disheartening to read such optimistic projections in a vacuum. True progress requires addressing systemic inequities alongside clinical advancements. Until then many will simply remain collateral damage of pharmaceutical evolution.
Sakshi Mahant
There is truth in your critique but national policies vary greatly by region. In India we are seeing more awareness regarding allopurinol reactions recently. Screening is becoming accessible in major cities though rural areas lag behind significantly. We must acknowledge the progress made while remaining humble about gaps. Collaboration across borders helps share data on rare adverse events effectively. Respecting local contexts allows for safer adoption of global standards.
HARSH GUSANI
Indian healthcare is fine why do you always talk about gaps ☽ Western drugs cause most harm anyway 🪭 Stop blaming our doctors for global issues 👄 We survive much harder problems than this daily 💪 You people just worry too much about skin peeling 👄
Branden Prunica
I was reading through this and suddenly felt a chill down my spine. The thought of losing my skin feels like a horror movie coming to life. Imagine waking up raw and bleeding everywhere with nowhere to hide. Some days I think the body is more fragile than we ever give it credit for. We really walk on borrowed time with every single pill we swallow. Please take care of yourselves out there in this dangerous world. I am literally shaking just thinking about the hospital ward scenario described here. Stay safe friends this is genuinely terrifying information.