When you have an autoimmune disease and are thinking about having a baby, the questions don’t stop at "Can I get pregnant?" They quickly turn to: "Which of my meds are safe?" "What happens if I stop them?" "Will my baby be okay?" The fear isn’t irrational. Many women have been told to stop their meds before conception, only to end up with a flare that’s far more dangerous than the drugs themselves. But the science has changed - dramatically.
Most Medications Are Safer Than You Think
The old rule was simple: stop everything. But that’s not what the experts say anymore. In February 2025, the European Alliance of Associations for Rheumatology (EULAR) released updated guidelines after reviewing over 1,200 studies. Their conclusion? 87% of standard autoimmune medications can be safely continued through conception, pregnancy, and breastfeeding. That’s not a guess. It’s based on data from tens of thousands of pregnancies. Take hydroxychloroquine. It’s used for lupus and rheumatoid arthritis. Across more than 12,450 documented pregnancies, it showed a 98.7% safety rate with no increase in birth defects. In fact, women who stayed on it had 50% lower risk of preeclampsia and 66% fewer flares during pregnancy. Stopping it doesn’t protect the baby - it puts both mother and baby at greater risk. Azathioprine? Also safe. Over 5,800 pregnancies showed a 95.3% safety rate. And here’s the key point: the risk of preterm birth was 2.1% for women on azathioprine, but jumped to 8.7% for those whose disease flared because they stopped their meds. The disease itself is often the bigger threat.What About Biologics? TNF Inhibitors Are Still the Gold Standard
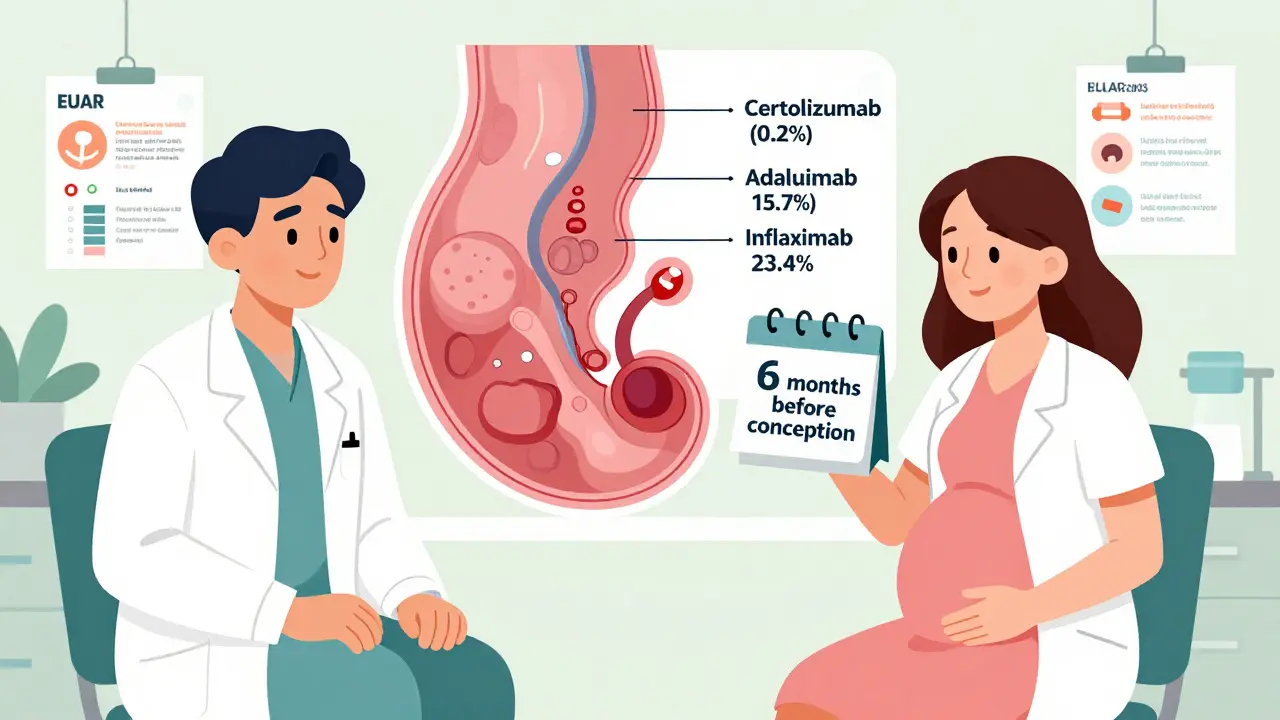
Tumor necrosis factor (TNF) inhibitors - drugs like adalimumab, infliximab, and certolizumab - are commonly used for rheumatoid arthritis, Crohn’s, and psoriasis. For years, people assumed they were risky. Now we know better. Across nearly 29,000 pregnancies, TNF inhibitors as a group had a 94.8% safety rate. But not all are the same. Certolizumab pegol stands out. It has the lowest placental transfer at just 0.2% of maternal blood levels. That means almost none reaches the baby. Adalimumab? About 15.7%. Infliximab? Up to 23.4%. This matters most in the third trimester, when the placenta becomes a more efficient transporter. That’s why many doctors now recommend switching to certolizumab if you’re planning to carry past 30 weeks. You don’t need to stop. You just need to choose the right one. And here’s something most OBs still don’t know: stopping TNF inhibitors at 32 weeks? No evidence supports it. A 2021 study of over 14,000 babies exposed to these drugs after 32 weeks found identical infection rates - 7.3% - compared to babies not exposed. The fear of newborn infection? Unfounded.These Medications Must Be Stopped - And How Far in Advance
Not all drugs are safe. Two are absolute red flags:- Methotrexate: Linked to 17.8% risk of major birth defects - including skull, spine, and limb abnormalities. It’s classified as Category X. You must stop at least 3 months before trying to conceive.
- Mycophenolate mofetil: Causes severe defects like cleft lip, ear abnormalities, and heart problems. FDA black box warning updated in March 2023. Requires a 6-week minimum washout, but experts recommend at least 3 months for safety.
What About Newer Drugs? JAK Inhibitors and Biosimilars
JAK inhibitors like tofacitinib and upadacitinib are newer, powerful options. But there’s a gap in data. EULAR recommends avoiding them entirely during pregnancy. The Japanese Rheumatism Society disagrees - their registry of 47 pregnancies showed only a 1.8% anomaly rate, close to the national average of 2.1%. That’s why some doctors in Japan allow upadacitinib through the first trimester. In the U.S., the advice is still "don’t use." The NIH launched a $12.7 million research network in January 2024 to study exactly this. Results won’t be in until 2029. Until then, caution wins. Biosimilars? No worries. Eight adalimumab biosimilars - like Amjevita and Hyrimoz - hit the market after Humira’s patent expired in January 2023. They’re not "weaker" versions. They’re exact copies. The FDA confirms they have the same safety profile during pregnancy. If your insurance switched you to a biosimilar, you’re still protected.Preconception Planning Isn’t Optional - It’s Critical
The biggest mistake women make? Waiting until they’re pregnant to talk about meds. That’s too late. The ideal window? At least 6 months before conception. That’s when your rheumatologist and maternal-fetal medicine specialist should sit down together. One reviews your disease activity. The other reviews fetal risks. A pharmacist helps time the switch. At Duke University’s pregnancy clinic, they cut high-risk medication use at conception from 38.7% to just 8.2% by making this a standard part of care. That’s not magic. It’s planning. A 2022 survey by the Lupus Foundation found women who had this kind of coordinated care had 53% fewer unplanned medication stops and 37% higher rates of full-term births. Real numbers. Real outcomes.What About Breastfeeding?
You don’t have to choose between your health and your baby’s nutrition. Over 98% of biologics transfer minimally into breastmilk. Adalimumab? Only 0.005% to 0.13% of maternal serum levels. That’s less than a drop. Most experts say it’s safe to continue all biologics while nursing. Hydroxychloroquine? Safe. Azathioprine? Safe. Even certolizumab? Safe. The risk of a flare from stopping your meds is far greater than any theoretical risk from the tiny amount in your milk.Real Stories, Real Consequences
One woman posted on MyHealthTeams: "Continued hydroxychloroquine throughout pregnancy - baby born at 39 weeks, 7 lbs 10 oz, zero complications." Simple. Clean. Safe. Another shared on HealthUnlocked: "Stopped adalimumab at 8 weeks because my OB said to. Flared badly at 20 weeks. Needed 20mg of prednisone. Developed gestational diabetes. Delivered at 34 weeks." The difference? One followed evidence. The other followed outdated advice. Reddit’s r/Autoimmune community has over 287,000 members. In 2023 alone, 1,247 posts described successful pregnancies on certolizumab. Only 218 mentioned traumatic experiences from mycophenolate exposure. The pattern is clear: informed choices lead to better outcomes.What’s Next? The Future Is Personalized
We’re moving beyond one-size-fits-all. Dr. Megan Clowse’s team at Duke developed a prediction tool that calculates your personal flare risk during pregnancy using 12 clinical factors - from disease duration to antibody levels. It’s 87.3% accurate. Soon, your care won’t be based on general guidelines. It’ll be based on you. EULAR is releasing a patient decision aid in November 2024. ACOG plans to update its guidelines in Q2 2025 to match EULAR’s 2024 data. The FDA’s new Pregnancy Exposure Registry Enhancement Program aims to cut the evidence gap for new drugs from 18-24 months down to 6-9. This isn’t just about drugs. It’s about control. You don’t have to choose between being healthy and having a baby. You just need the right information at the right time.Can I get pregnant if I have lupus or rheumatoid arthritis?
Yes. Most women with autoimmune diseases can have healthy pregnancies. The key is disease control before conception. Flares during pregnancy raise risks for preterm birth, preeclampsia, and low birth weight. With proper planning and medication management, live birth rates are comparable to the general population.
Is hydroxychloroquine safe during pregnancy?
Yes. Hydroxychloroquine is one of the safest autoimmune medications for pregnancy. It’s been used in over 12,450 pregnancies with no increase in birth defects. It also reduces lupus flares by 66% and cuts preeclampsia risk by half. It’s recommended to continue throughout pregnancy and even while breastfeeding.
Should I stop my biologics during pregnancy?
Not necessarily. TNF inhibitors like certolizumab pegol, adalimumab, and etanercept are safe to continue. Certolizumab is preferred in the third trimester due to minimal placental transfer. Stopping these drugs increases your chance of a flare by 3 to 5 times - which is far riskier than continuing them.
What if I got pregnant while on methotrexate?
Contact your rheumatologist and OB immediately. Methotrexate is teratogenic and carries a 17.8% risk of major birth defects. However, not every exposure leads to problems. An ultrasound and genetic counseling can help assess fetal risk. Going forward, methotrexate must be stopped at least 3 months before any future attempts at conception.
Can I breastfeed while on autoimmune medication?
Yes. Over 98% of biologics and DMARDs transfer negligibly into breastmilk. Adalimumab, certolizumab, azathioprine, and hydroxychloroquine are all considered safe. The risk of a disease flare from stopping treatment is much greater than any risk to your baby from the tiny amount of drug in your milk.
When should I start planning for pregnancy?
At least 6 months before you plan to conceive. This gives time to switch unsafe drugs (like methotrexate or mycophenolate) to safer alternatives, stabilize your disease activity, and coordinate care between your rheumatologist and maternal-fetal specialist. Waiting until you’re pregnant increases the risk of complications for both you and your baby.
Are biosimilars safe during pregnancy?
Yes. Biosimilars like Amjevita and Hyrimoz are exact copies of their reference biologics (e.g., Humira). The FDA confirms they have identical safety profiles during pregnancy. If your insurance switched you to a biosimilar, you are not at increased risk - and you’re still protected.
What to Do Next
If you’re thinking about pregnancy:- Talk to your rheumatologist now - not when you’re pregnant.
- Ask if your meds are on the safe list. If you’re on methotrexate or mycophenolate, ask for a switch plan.
- Request a joint visit with a maternal-fetal medicine specialist.
- Use tools like the MotherToBaby registry or the Lupus Foundation’s resources to track your meds.
- Don’t rely on outdated OB advice. The guidelines changed in 2025. Your care should too. This isn’t about fear. It’s about control. You can have a baby. You can stay healthy. You just need the right plan - and the right information.





Milad Jawabra
I’ve been on hydroxychloroquine for 8 years and just had my second kid. No issues. Zero. My OB was freaking out until I showed her the EULAR guidelines. She apologized. Honestly? If you’re scared to stay on your meds, you’re listening to the wrong people. Stop letting fear dictate your health. 🤝
Shivam Pawa
Data says 87 percent safe but still i think body knows best. some women feel different. not all flares are equal. maybe the system is too rigid. science is not god. just data.
Donna Zurick
This is the kind of info that changes lives. I wish my rheum had told me this 2 years ago when I was panicking about stopping my meds. Now I’m 32 weeks pregnant on certolizumab and my baby’s kicking like a soccer player. Thank you for sharing the real numbers. 💪
Tobias Mösl
87% safe? That’s a lie wrapped in a statistic. Pharma owns EULAR. Look at the funding. Who paid for these ‘studies’? Big pharma. The FDA has been compromised since 2018. They want you dependent. They want you pregnant on drugs so you keep buying. Certolizumab? It’s a Trojan horse. Wait till your kid is 5 and has autism. Then you’ll see. 🤡
tatiana verdesoto
I’m so glad this exists. I had my first pregnancy on methotrexate. We lost the baby at 10 weeks. No one told me. No one warned me. I cried for months. If this post had been around then… I wouldn’t have blamed myself. You’re not alone. And you don’t have to suffer in silence. Reach out. Ask. Demand better.
Ethan Zeeb
I’m a nurse in a maternal-fetal unit. We used to tell women to stop everything. Now we have a protocol. We screen at 6 months out. We map meds. We coordinate. The drop in preterm births? Real. The drop in flares? Real. This isn’t theory. It’s daily practice. Stop asking if it’s safe. Start asking how to do it right.
Darren Torpey
Let me paint you a picture: You’re not choosing between a pill and a baby. You’re choosing between a flare and a baby. One’s a slow burn. The other’s a lightning strike. Hydroxychloroquine? It’s your body’s bodyguard. Azathioprine? Your peacekeeper. Certolizumab? Your silent ninja. Don’t disarm yourself. You’re not a patient. You’re a warrior. And warriors don’t surrender before the battle.
Lebogang kekana
I’m from South Africa. We don’t have access to half these drugs. My rheum doc told me to stop everything. I did. Flared. Got hospitalized. Lost 3 months of work. My kid’s fine. But I wasn’t. If this info was available here? I wouldn’t have suffered. This is why we need global equity in care. Not just data. Access.
Jessica Chaloux
I’m so angry. I stopped my meds because my OB said "better safe than sorry." I flared. My baby was born at 31 weeks. I’m still in therapy. I hate myself. I hate the system. I hate that I trusted someone who didn’t know better. I hope this post saves someone else. Please. Don’t be me.
Mariah Carle
We are not merely biological vessels. We are the intersection of chemistry, identity, and will. To fear a drug is to fear the self. The body does not lie. The system does. The truth is not in the guideline - it is in the lived experience. And if you are reading this… you are already choosing your truth. 🌌